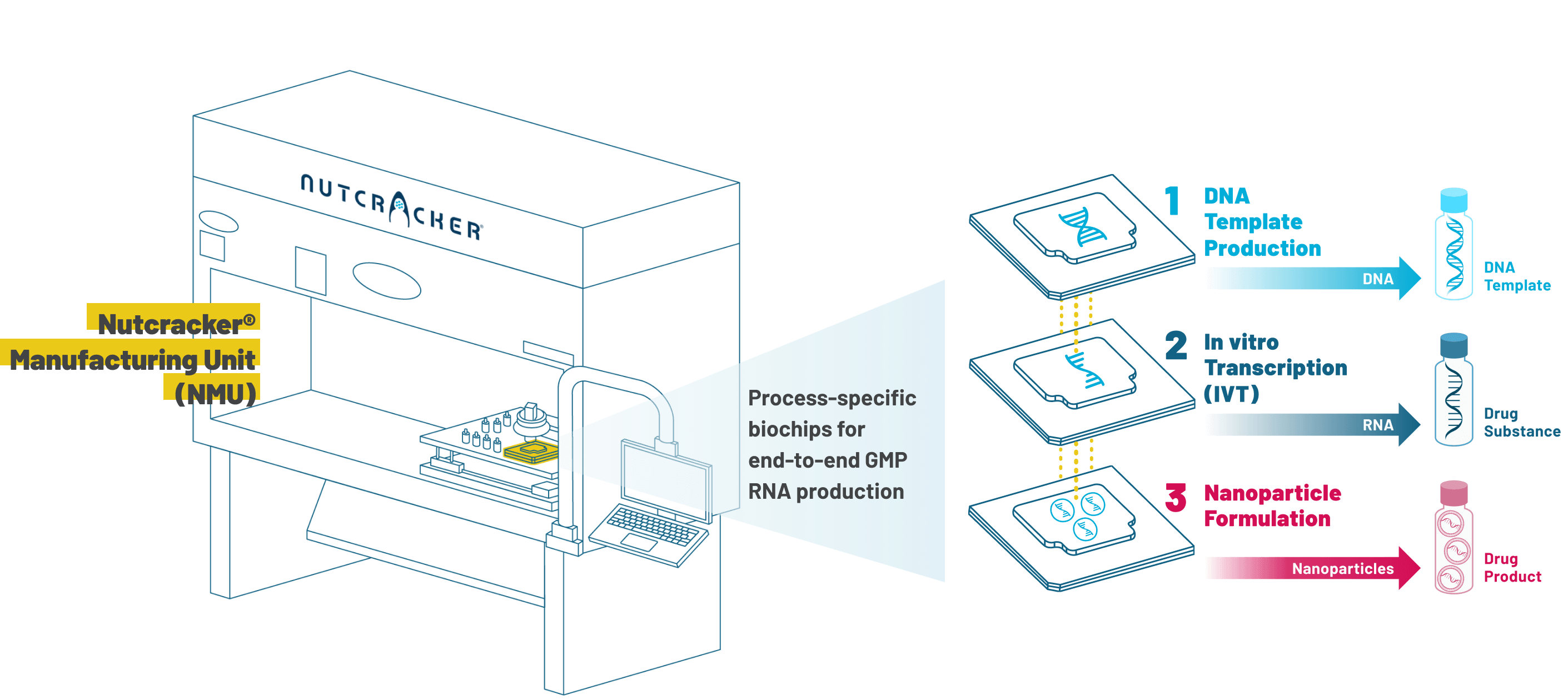
Engineering meets biology in the Nutcracker® Manufacturing Unit (NMU)
Realizing the promise of RNA therapeutics requires increasing the speed and scale at which they can be discovered, developed, and manufactured. The NMU compresses an entire manufacturing facility into an office printer-sized box that takes a DNA sequence all the way to a fully formulated mRNA therapy.
At the heart of the NMU is the biochip, which leverages microfluidics to fit the entire function of a bioreactor and purification column onto a small, three-inch square. Miniaturizing production with in-process monitoring enables tight control over all the reactions, reagents, and purifications for each step of the RNA manufacturing process.
Scalable microfluidics
Nutcracker Therapeutics’ proprietary biochips utilize a fully enclosed microfluidic path that requires minimal hands-on time. Fluids move across multiple chambers to the site of each reaction in a semi-continuous process that enables precise control, reproducibility, and scalability. Process-specific to template DNA production, in vitro transcription (IVT), or nanoparticle formulation, our biochips support research, clinical, and commercial use.

In-process biochip monitoring
Our consumable biochips with integrated process control features are the core of the NMU. Proprietary active monitoring and novel closed-loop process measurements are built directly into our biochips, and designed to ensure quality and consistency across every manufacturing cycle.

GMP-in-a-box
The automated processes of the NMU are tightly controlled by proprietary integrated software. Our systems and processes are designed to fully incorporate the controls required for GMP manufacturing, resulting in the highly consistent production of every lot of material.
Rapid cycle times
The automated nature of the NMU and the biochip’s ability to begin a new cycle before the previous cycle has been completed make it ideal for rapid iteration in the discovery and development of new therapeutics. The NMU is compatible with both off-the-shelf and patient-specific RNA formulations. The NMU can go from a patient sample to a personalized drug product in as little as a few weeks.

The art and science of RNA design
Nutcracker Therapeutics’ proprietary, cloud-based CodonCrackerSM software rapidly optimizes RNA sequences to fine-tune protein expression levels and manufacturability. Our RNA designs also incorporate novel sequence elements to enhance the activity of our products.

Novel delivery vehicles
Nutcracker Therapeutics’ nanoparticle technology offers a growing library of unique and effective Nutshell® delivery vehicles, including thousands of formulations that have been evaluated across more than 200 compounds. Nutshell delivery vehicles use peptoids – synthetic biochemicals that look and act like lipid nanoparticles but offer several advantages, including tunable chemical structures, uniform nanoparticle size, and low toxicity and immunogenicity.
See our platform in action

